Nuclear envelope,
Telomeres and DNA repair
IntGen Team : Integrity of genome
In a cell nucleus, chromosomes contain the genetic information, the genes, which code for the proteins. These are needed to operate the cell and the organism as a whole. Once a chromosome is damaged, the genes located on this chromosome can be altered, and the stability of the genome can be affected. The proteins encoded by these genes can be mutated or truncated, they are no longer able to fulfil their function. The damaged chromosome is signalled to the cell by protein molecular machineries, its repair is handled by other molecular machineries that may be specific to particular damage or breaks of one or both strands of DNA. Both damage signalling and repair involve a network of interactions between proteins.
In our team, we specifically study the molecular machineries involved in the networks that control the stability of the genome, through the signalling and repair of DNA damage, or the assembly and structure of the nuclear envelope. In order to understand how these molecular machineries function at the atomic level, we study the three-dimensional structure of the proteins and/or nucleic acids that compose them, and characterize their assembly and dissociation mechanisms.
Most of our studies result from tight collaborations with cell biologists and clinicians involved in characterizing the function of protein networks. When the molecules we study are involved in genetic diseases, we also aim at characterizing the many mutants detected by geneticists, in order to predict whether these mutants trigger the disease. The clinicians can then decide if family members of the patient presenting that same mutation also need a close follow-up.
For determining the three-dimensional structures of these complexes, we use an integrative structural biology approach that includes Nuclear Magnetic Resonance, X-ray crystallography and electron microscopy.
The composition and molecular architecture of the investigated complexes evolve throughout their action in the cell. In addition, most of the proteins involved in these complexes consist of several structural domains presenting distinct biological functions and linked together by less structured regions. This facilitates the coordination of their different biological functions. Our team aims at identifying high or low affinity interactions between protein domains, characterizing their mode of interaction with atomic resolution and elucidating their regulation modes, using biochemical and biophysical techniques (in solution, AUC, ITC, MST, SEC-MALS; and on biochips, BLI, SPR, switchSENSE) and Nuclear Magnetic Resonance methods. The impact of post-translational modifications on the three-dimensional structure and binding properties of partially flexible proteins is studied in vitro and in cell extracts using Nuclear Magnetic Resonance. The presence of complexes in the cell and their functional role are systematically addressed through collaborations with cell biologists and/or geneticists working on the signaling and repair of DNA damage and the organization of the nucleus.
Research themes
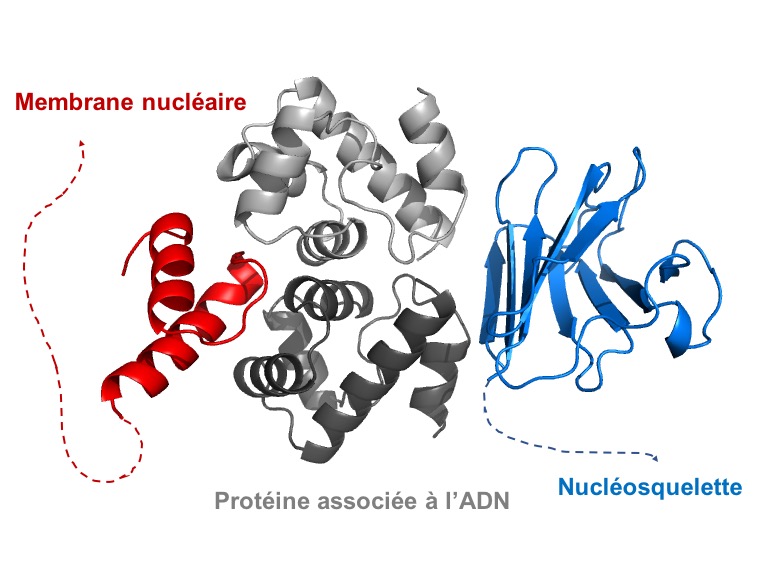
The nuclear envelope proteins, including lamins
(Sophie Zinn-Justin)
The nucleoskeleton is formed by a network of lamin filaments. We seek to understand how these lamins interact with proteins anchored at the nuclear membrane and chromatin, in order to contribute to the mechanical properties of the nucleus and to the stability of the genome. We are also studying the impact of mutations associated with genetic diseases (muscular dystrophies, premature aging syndromes) on the 3D structure and binding properties of these lamins (Samson, NAR 2018; Essawy, Cells 2019; Marcelot, FEBS J 2020).
Homologous recombination and BRCA1 / BRCA2 proteins
(Sophie Zinn-Justin)
Repair of double-strand breaks can be achieved by homologous recombination in S and G2 phase, once the DNA has been replicated and an intact and spatially close copy of the sister chromatid is available. Two essential proteins of this repair pathway, BRCA1 and BRCA2, are mutated in hereditary breast and ovarian cancers. These proteins have large unstructured regions and we characterize their binding properties: 3D structures of the complexes, regulation of these interactions by phosphorylation events (Petitalot, Mol Cancer Res 2019; Ehlen, Nat Commun 2020).
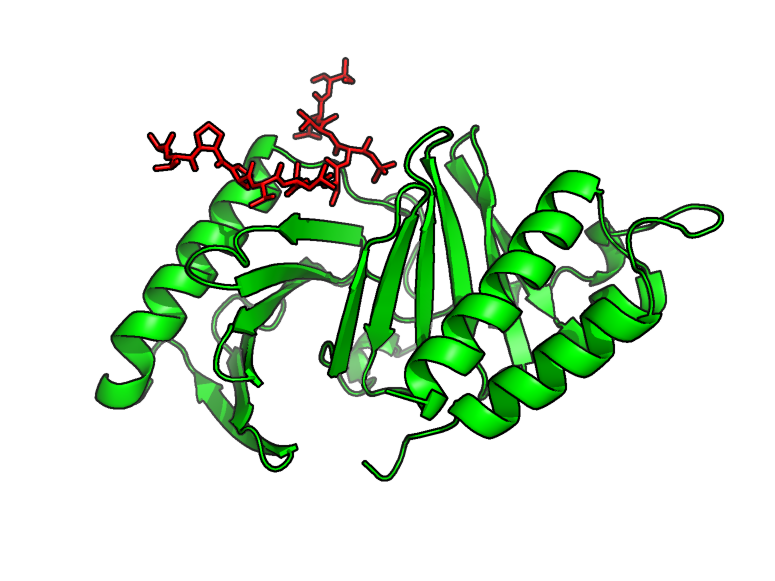
Non-homologous recombination (NHEJ)
(Jean-Baptiste Charbonnier, Virginie Ropars)
Double-strand breaks in DNA are the most toxic damage. They are mainly treated by the NHEJ pathway in humans. We are studying the 3D structures of major assemblies of this pathway (e.g. XRCC4-XLF (Ropars, PNAS, 2011), Ku-DNA-XLF (Nemoz, Nat Struct Mol Biol, 2018)) by crystallography and cryoEM.
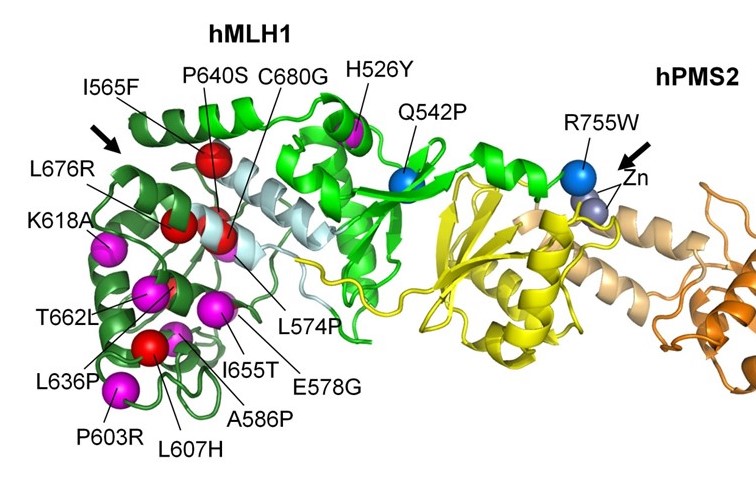
The MMR
(Jean-Baptiste Charbonnier)
MutL’s eukaryotic counterparts are central actors in the repair pathway of mismatches but also in mitotic and meiotic recombination. We study these multiple functions through structural and functional studies (MutLα (Gueneau, Nat Struct Mol Biol, 2013) and MutLγ (Cannavo, Nature, 2020, Sanchez, PNAS, 2020).
Damage signalling
(François-Xavier Theillet)
DNA damage activate kinases that trigger repair or cell-cycle arrest by phosphorylating a number of targets. Among them, we study the DNA-PK-Chek2:Wip1:Mdm2:p53 network, whose regulation by phosphorylation determines cell fate (Alik et al. 2020). We also develop methods to observe kinases in-cell at the atomic scale (Theillet et al. Nature 2016).
Our approaches
Nuclear Magnetic Resonance
Our team is responsible for the NMR platform at I2BC (link), which is used to characterize the solution structures, modifications and interactions of our proteins of interest. We also perform studies for other teams at I2BC, in particular the teams of the Virology department (Vernhes, Sci Rep 2017; Chaban, PNAS 2015).
Crystallography
We routinely use biological crystallography in the laboratory. Our team works in tight collaboration with beamline scientists at the SOLEIL synchrotron to determine crystal structures.
Electron microscopy
We study proteins or large assemblies by cryoEM. We use several platforms including those of I2BC and the Institut Pasteur and we collaborate with the University of Cambridge. We also collaborate with G Belot for the use of Origami for cryoEM.
Molecular modeling
We determine the 3D structure of proteins and biomolecular complexes from heterogeneous data (NMR, SAXS, Cryo-EM, mutagenesis… ; Cuniasse Curr Opin Struct Biol, 2017). We also perform Molecular Dynamics simulations. All these techniques were recently applied to proteins in the field of virology and genome integrity.
Biophysical characterization of protein interactions
(Paloma Fernández-Varela)
Our team offers access to an innovative method for measuring protein interactions, the switchSense, which uses oligonucleotides as interaction sensors (DRX2 instrument, DBS).
Protein Expression in
eukaryotic systems
Our team is responsible for the insect cells expression facility of the I2BC.
Teaching
Live classes at the Paris-Saclay University, The University of Technology of Compiègne,
E-learning : Production of the MOOC « Voyage au cœur du vivant avec des rayons X : la cristallographie. » (Marie-Hélène Le Du), broadcasted on France Université Numérique (https://www.fun-mooc.fr/). MOOC videos are also available on the Youtube channel: https://bit.ly/2FsAiDa
Workshops :
- Training School « Macromolecular interactions in vitro, comparing classical and innovative approaches (Paloma Fernández Varela) : https://mosbio.sciencesconf.org/ ;
- Biocrystallography workshop in collaboration with SOLEIL synchrotron (March 2016, April 2018) (Marie-Hélène Le Du) ;
- Ecole Nationale de Biologie Structurale Intégrative, held once a year since 2014. (Marie-Hélène Le Du).
- Protein interactions : in vivo, in vitro and in silico approaches, PhD workshop ED569 Innovation Therapeutique (since 2007)
- « Post-translational Modifications », Protein Biochemistry courses, Master 2 Institut Pasteur/Sorbonne Université.
team

Group Leader Researcher

Group Leader Researcher

Researcher

Researcher
Researcher

Researcher

Researcher
Engineer

Engineer assistant
Engineer assistant

Technician

Engineer

PhD student
Master Student
Master Student

PhD student

PhD student
PhD student

PhD student
PhD student
Our main collaborators
- (NHEJ) JP de Villartay (Inst Imagine, Paris), P Calsou (IPBS, Toulouse), M Modesti (CRCM, Marseille), M Delarue (Inst Pasteur, Paris), T Blundell (Univ Cambridge, UK), E Rothenberg (NY Univ, USA), T Strick (ENS, Paris) M Betermier (I2BC, Gif)
- (MMR) V Borde (I Curie, Paris), P Cejka (FBS, Switzerland), S Takeda (University of Shenzhen, Japan) A Satori (IMCR, Zurich)
- (lamins) HJ Worman (Columbia Uni, NY, USA), B. Buendia (Uni Paris Diderot)
- (RH) A Carreira (Institut Curie, Orsay), S Caputo (Institut Curie, Paris)
- (new techniques) A. Lange (FMP Berlin, Allemagne), G Belot (CBS, Montpellier)
Main publications
Ghouil, Rania, Simona Miron, Lieke Koornneef, Jasper Veerman, Maarten W. Paul, Marie-Hélène Le Du, Esther Sleddens-Linkels, et al. 2021. “BRCA2 Binding through a Cryptic Repeated Motif to HSF2BP Oligomers Does Not Impact Meiotic Recombination.” Nature Communications 12 (1): 4605. https://doi.org/10.1038/s41467-021-24871-6.
Dai, Jingqi, Aurore Sanchez, Céline Adam, Lepakshi Ranjha, Giordano Reginato, Pierre Chervy, Carine Tellier-Lebegue, et al. 2021. “Molecular Basis of the Dual Role of the Mlh1-Mlh3 Endonuclease in MMR and in Meiotic Crossover Formation.” Proceedings of the National Academy of Sciences of the United States of America 118 (23): e2022704118. https://doi.org/10.1073/pnas.2022704118.
Marcelot, Agathe, Ambre Petitalot, Virginie Ropars, Marie-Hélène Le Du, Camille Samson, Stevens Dubois, Guillaume Hoffmann, et al. 2021. “Di-Phosphorylated BAF Shows Altered Structural Dynamics and Binding to DNA, but Interacts with Its Nuclear Envelope Partners.” Nucleic Acids Research 49 (7): 3841–55. https://doi.org/10.1093/nar/gkab184.
Ehlén Å, Martin C, Miron S, Julien M, Theillet FX, Ropars V, Sessa G, Beaurepere R, Boucherit V, Duchambon P, El Marjou A, Zinn-Justin S, Carreira A. Proper chromosome alignment depends on BRCA2 phosphorylation by PLK1. Nat Commun. 2020 Apr 14;11(1):1819.
Alik A, Bouguechtouli C, Julien M, Bermel W, Ghouil R, Zinn-Justin S, Theillet FX. Sensitivity-Enhanced 13 C-NMR Spectroscopy for Monitoring Multisite Phosphorylation at Physiological Temperature and pH. Angew Chem Int Ed Engl. 2020 Jun 22;59(26):10411-10415. doi: 10.1002/anie.202002288.
Nemoz C, Ropars V, Frit P, Gontier A, Drevet P, Yu J, Guerois R, Pitois A, Comte A, Delteil C, Barboule N, Legrand P, Baconnais S, Yin Y, Tadi S, Barbet-Massin E, Berger I, Le Cam E, Modesti M, Rothenberg E, Calsou P, Charbonnier JB. XLF and APLF bind Ku80 at two remote sites to ensure DNA repair by non-homologous end joining. Nat Struct Mol Biol. 2018 Oct;25(10):971-980. doi: 10.1038/s41594-018-0133-6.
Gaullier G, Miron S, Pisano S, Buisson R, Le Bihan YV, Tellier-Lebègue C, Messaoud W, Roblin P, Guimarães BG, Thai R, Giraud-Panis MJ, Gilson E, Le Du MH. A higher-order entity formed by the flexible assembly of RAP1 with TRF2. Nucleic Acids Res. 2016 Feb 29;44(4):1962-76. doi: 10.1093/nar/gkv1531.
Theillet FX, Binolfi A, Bekei B, Martorana A, Rose HM, Stuiver M, Verzini S, Lorenz D, van Rossum M, Goldfarb D, Selenko P. Structural disorder of monomeric α-synuclein persists in mammalian cells. Nature. 2016 Feb 4;530(7588):45-50. doi: 10.1038/nature16531.
For all the publications of the Team click on the button below.
External funding

INCA

Sanofi CIFRE

