Light Energy Conversion
The Light Energy Conversion (LEC) team aims to understand how photosynthetic systems control energy flow and to translate these principles into bio inspired approaches for solar energy harvesting. We investigate the factors that regulate ultrafast excitation energy transfer and charge separation across multiple length scales, from membrane level reorganisation to pigment level interactions. This integrated approach provides a global view of the natural strategies employed to regulate the energy flow. We translate those principles to develop bio-inspired artificial systems capable of effectively collecting solar energy.
Architecture and Structural Plasticity of Photosynthetic Membranes
Photosynthetic organisms rely on several regulatory mechanisms to control energy flow and remain efficient under fluctuating conditions. To understand regulation at mesoscopic scales, we characterise the architectural plasticity of chloroplasts and thylakoid membranes by mapping how they reorganise in response to external stimuli. We build functional 3D maps of intact photosynthetic membranes by using chlorophyll fluorescence as a direct readout of structural changes. By exploiting the natural diversity of photosynthetic membrane architectures across cyanobacteria, algae, and plants, we connect organisation to function. As an example, we reconstruct photosystem I and II topology and track how it responds to changes in light intensity and light quality to identify both shared and organism specific regulatory strategies. Using cyanobacteria as a model system, the same 3D mapping approach also can be employed to explore the links between antagonistic processes like O2 production by PSII and respiration. These studies rely on microscopy methods developed in-house for high resolution fluorescence and Raman imaging. Our approach resolves photosynthetic membrane autofluorescence at about 150 nm laterally and about 300 nm axially, strongly limiting out of focus contamination and complementing other techniques that study in vivo membrane architectures.
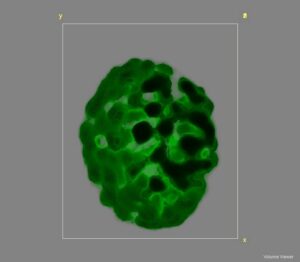
Figure 1: 3D reconstruction of an Arabidopsis thaliana chloroplast using chlorophyll fluorescence.
Structure and Fate of Excited States in Natural Photosynthesis
Harvesting sunlight is essential for the bioenergetic balance of photosynthetic organisms. Their photosynthetic apparatus captures solar photons and transfers excitation energy to reaction centres, where it is converted into chemical potential energy with very high efficiency under optimal conditions. In natural environments, however, light intensity and spectral quality fluctuate continuously, so photosynthetic systems must respond rapidly to avoid photodamage. The photosynthetic apparatus therefore combines highly efficient light harvesting with the rapid activation of photoprotective dissipation when excess excitation builds up. However, the detailed molecular mechanisms that underpin both near unity harvesting efficiency and effective photoprotection remain incompletely understood. Our work aims to understanding these molecular mechanisms by characterising the key excited states involved in energy transfer and quenching. One of the keys to understand those mechanisms is in resolving the structure of the excited states involved in these processes, their nature and reactivity. Our laboratory is part of a large-scale, international project to determine whether photosynthetic energy transfer exhibits a quantum character – does the system explore more than one transfer pathway at the same time? – as well as whether vibrational coupling aides the transfer process. We study light harvesting systems from purple bacteria, algae, and higher plants, ranging from isolated pigment protein complexes to more intact assemblies, to identify how pigment organisation and vibrational structure regulate energy flow. To access excited state structure directly, we rely on vibrational spectroscopies. Our laboratory is internationally recognised for resonance Raman and related vibrational methods. We have recently installed a state-of-the-art femtosecond stimulated resonance Raman instrument, which provides highly selective vibrational fingerprints of excited states with femtosecond time resolution.
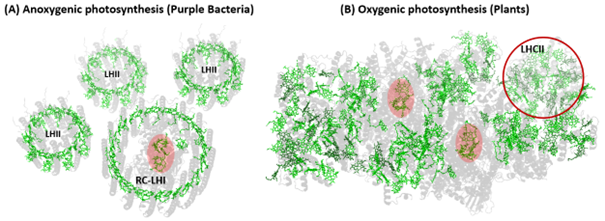
Figure 2| Structural comparison of anoxygenic and oxygenic photosynthetic protein supercomplexes.
Bio-inspired artificial photosynthetic systems
The current model for energy production relies on fossil fuels, which are a limited resource and generate CO2, a greenhouse gas that induces perturbations of the planet’s climate. Through photosynthesis, solar radiation provides the energy to power the biosphere. However, the high level of efficiency observed for biological photosynthesis has yet to be even approached in artificial systems. The remarkable sophistication of natural photosynthetic machinery, which converts solar energy through hierarchies of pigments, proteins and membranes, stands in sharp contrast to the still relatively rudimentary designs of current photovoltaic and photocatalytic technologies. By understanding how photosynthesis manages light harvesting and energy conversion, we extract design principles that can inspire cleaner and more effective solar energy technologies. One major focus in our team is singlet fission, a process in which one absorbed photon can generate two long lived triplet excitations, with a theoretical quantum efficiency of 200%. In chromoplasts purified from flowers and fruits, we observed that when carotenoids assemble into disordered aggregates, the triplet states formed ultrafast by singlet fission live longer than in well ordered crystals. This extended lifetime is important because it increases the time window for photochemistry. Inspired by chromoplast carotenoid assemblies, we design perylene based chromophores, which are absent from Nature but combine singlet fission capability with higher oxidation potentials and improved photostability. In water soluble perylene assemblies, we created a dynamic and disordered energetic landscape that promotes triplet separation and slows recombination into the nanosecond range, reaching triplet yields around 180%. Across these systems, we address a practical question: which molecular arrangements favour triplet formation, charge transfer, and minimal recombination? We tackle this using state selective vibrational spectroscopy, in particular femtosecond stimulated resonance Raman, which tracks transient species with structural specificity and femtosecond time resolution. This mechanistic understanding supports the next step of our programme: coupling chromophore pairs to catalytic centres to drive multi electron oxidation reactions.
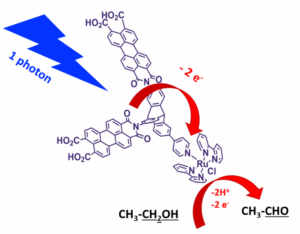
Figure 3| Molecular representation of a bio-inspired antenna–catalyst dyad consisting of two perylene chromophores covalently linked to a ruthenium molecular catalyst
team

Group Leader Researcher

Researcher

Researcher

Researcher

Researcher

Researcher

Engineer
Technician
PhD student
PhD student
Some publications
2026
Bercy, R., V. D’mello, A. Gall, C. Ilioaia, A.A. Pascal, J.J. Romero, B. Robert, and M.J. Llansola-Portoles, Reassessing carotenoid photophysics – new light on dark states. arXiv:2601.00316v1, 2026.
Banos-Gutierrez, J., R. Bercy, Y.G. Jomaso, S. Balci, G. Pirruccio, J.H. Stenlid, M.J. Llansola-Portoles, and D. Finkelstein-Shapiro, Molecular structure, binding, and disorder in TDBC-Ag plexcitonic assemblies. arXiv:2601.22022, 2026.
2025
Maillot, B., H. Rashid, R. Bercy, J.F. Audibert, M.J. Llansola-Portoles, I. Leray, F. Miomandre, and V. Brasiliense, Nanometrology assisted chemical fabrication: direct laser writing of porphyrins onto complex surfaces. Nanoscale, 2025. 17(33): p. 19220-19230.
Magne, C., V. Veremeienko, R. Bercy, M.-H. Ha-Thi, A.A. Arteni, A.A. Pascal, M. Vengris, T. Pino, B. Robert, and M.J. Llansola-Portoles, Singlet fission in heterogeneous lycopene aggregates. Scientific Reports, 2025. 15(1): p. 5593.
External funding


