Cytometry
Equipments
All the equipments are accessible after training by the facility engineers.

Year of commissioning : 2017
Lasers:
- 405 nm (4 detectors)
- 488 nm (2 detectors)
- 561 nm (4 detectors)
- 640 nm (3 detectors)
Holder:
- 5-ml tubes
- Eppendorf 2-ml tubes
- Eppendorf 1.5-ml tubes
- 96-wells plate
Up to 10,000 events per second
Software: CytExpert 2.5
Optical diagram available here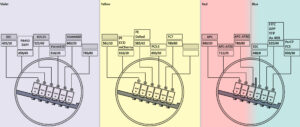

Year of commissioning : 2011
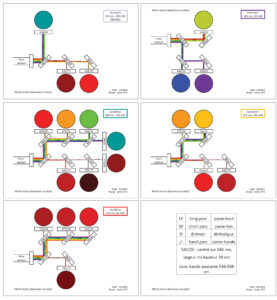
Lasers:
- 355 nm (3 detectors)
- 405 nm (2 detectors)
- 488 nm (6 detectors)
- 561 nm (3 detectors)
- 640 nm (4 detectors)
Holder:
- 50-ml tubes
- 15-ml tubes
- 5-ml tubes
- Eppendorf 2-ml tubes
- Eppendorf 1.5-ml tubes
Ways of sorting:
- 1.5 ml to 5 ml tubes => 6ways
- 15 ml to 50 ml => 2 ways
- 6 to 96 wells plate => 1way
Up to 35,000 events per second
Software: Summit 6.3
Optical diagram available here
Available software
Analysis station with the following software:
![]() Flowlogic 7.2 (Miltenyi-Biotech / lnivai)
Flowlogic 7.2 (Miltenyi-Biotech / lnivai)
For the analysis of flow cytometry data with 3 modules:
Analysis (gateLogic): overlays, heat maps, cell cycle, kinetics, cell proliferation
Statistics (graphLogic): statistical analyzes and creation of graphs
Report (doclogic): layout, pdf generation
 Kaluza Analysis Software (Beckman Coulter)
Kaluza Analysis Software (Beckman Coulter)
Complete cytometry software, with the possibility of exporting fcs data to csv
Adequate software for making high resolute plots (300 dpi) for publication
CytExpert 2.5 (Beckman Coulter, available in I2BC self-service)
Basic and free software that controls the Cytoflex

Summit 6.3 (Beckman Coulter, available in I2BC self-service)
Basic and free software that controis the sorter
![]() R and Bioconductor tools (available in I2BC store)
R and Bioconductor tools (available in I2BC store)
It is possible to have personal script for data analysis
Rates
Training

Imagerie-Gif Cytometry facility proposes training on all its equipment all along the year.
Specific training are proposed once a year to blow your mind with flow cytometry : See here
Services
All the equipment are accessible after training by the facility’s engineers.
The facility engineers can perform experiments for the external users.
When the implication of the engineer is consistent, it has to be included in the authorship of the publication.
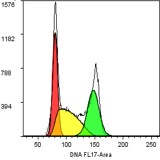 Determination of the quantity of nuclear DNA for the study of cell cycles and endoreplication.
Determination of the quantity of nuclear DNA for the study of cell cycles and endoreplication.
It is possible to calculate cell proportion in G1-, S- and G2/M-phase of cell cycle. You can also measure other fluorescence parameters and follow them during cell cycle.
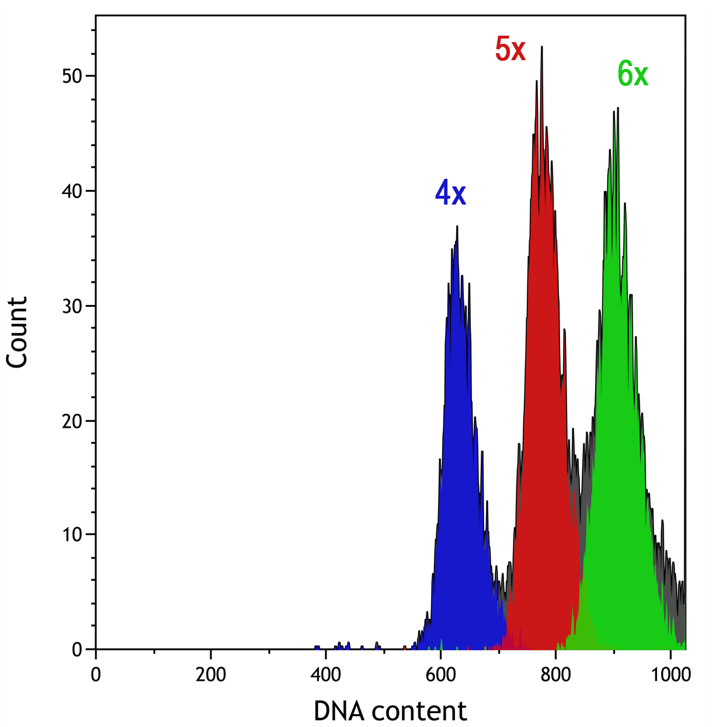
DNA assay for ecological and systematic research and variety improvement (ploidy analysis).
It is simple to measure genome size of plants or insects with cytometry by isolating nuclei mechanically in adequate buffer.
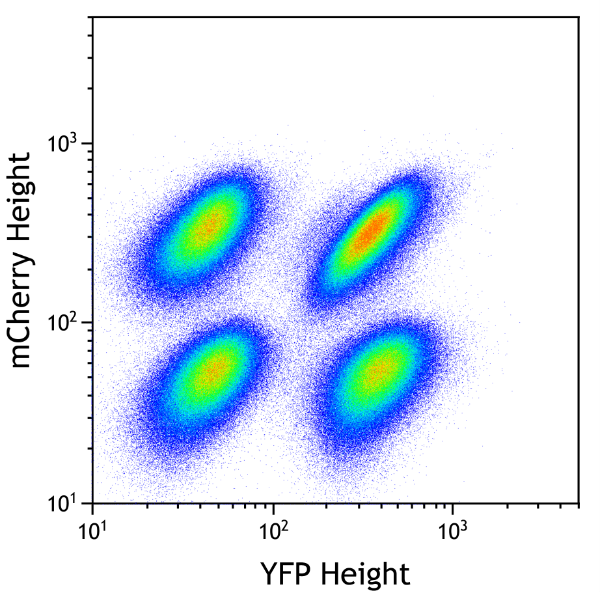 Monitoring of gene activity by expression of a reporter gene (such as that of “Green Fluorescent Protein” -GFP or other fluorescent proteins).
Monitoring of gene activity by expression of a reporter gene (such as that of “Green Fluorescent Protein” -GFP or other fluorescent proteins).
You can measure percent of cells positive for one or more fluorescent protein expressed by your cells. You can also compare the fluorescence intensity, reflecting expression level of your protein.
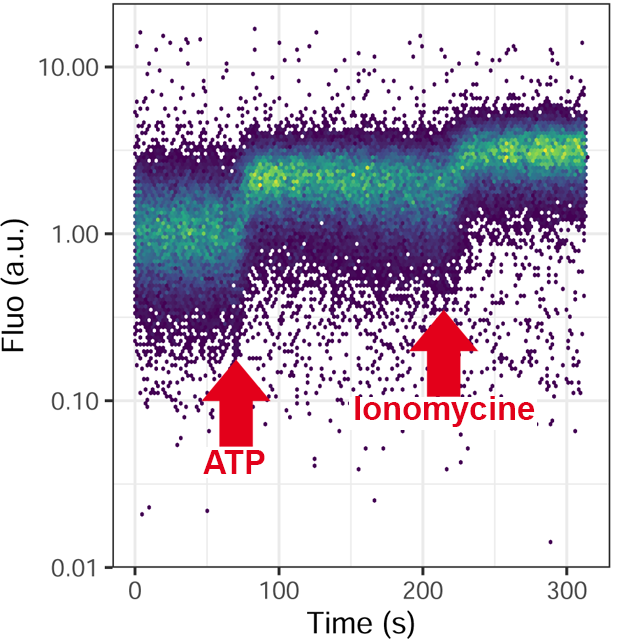 Measurements of the metabolic activities of the cell: dosage of calcium, pH, membrane potential, oxidative burst, glutathione …
Measurements of the metabolic activities of the cell: dosage of calcium, pH, membrane potential, oxidative burst, glutathione …
With appropriate biosensor, you can measure a metabolic activity by analyzing fluorescence kinetic in live after addition of drugs.
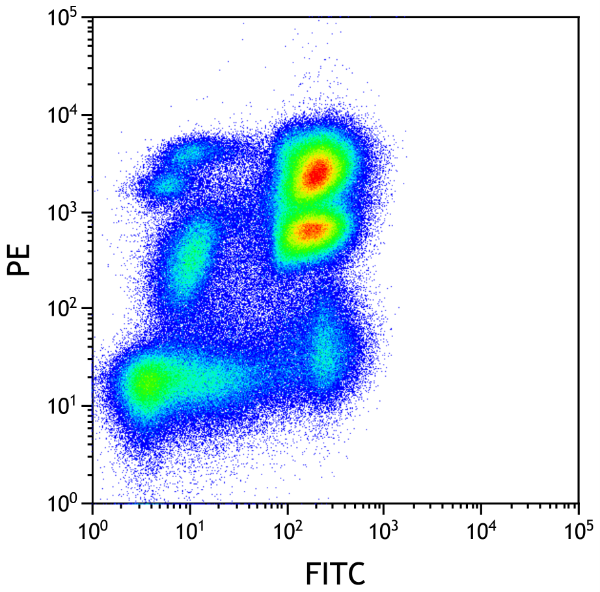 Immunological analyzes.
Immunological analyzes.
By choosing wisely your antibodies cocktail, you can measure several fluorescence parameters to identify a target population.
 Sorting of animal cells, protoplasts, yeasts, bacteria and cell organelles.
Sorting of animal cells, protoplasts, yeasts, bacteria and cell organelles.
Identifying your cell or organelle populations according to morphology and/or fluorescence allows you to sort and purify them in tubes (from Eppendorf 1.5 ml to Falcon 50 ml) or plates (6 to more than 96 wells).
Contact
Plateforme de Cytométrie / Cytometry Facility
Université Paris-Saclay, CEA, CNRS, Institute for Integrative Biology of the Cell (I2BC)
1 Rue de la Terrasse
91198, Gif-sur-Yvette, France.
Bourge Mickael, Head of facility, Researcher Engineer
Mail : mickael.bourge@i2bc.paris-saclay.fr
Phone: +33 1 69 82 46 30
Madiona Karine, Engineer
Mail : karine.madiona@i2bc.paris-saclay.fr
Phone: +33 1 69 82 46 30
Acknowledgements
Funding is essential for the proper functioning and development of the Imagerie-Gif facilities. To this end, it is essential to acknowledge the facility as soon as you have benefited from the help of the engineers or any equipment.
Use this sentence to acknowledge the Cytometry Core Facility :
The present work has benefited from Imagerie‐Gif core facility supported by I’Agence Nationale de la Recherche
(FBI ANR-24-INBS-0005 (BIOGEN); SPS ANR-17-EUR-0007, EUR SPS-GSR )
Use this address for the Cytometry Core Facility :
Cytometry Facility, Imagerie-Gif, Université Paris-Saclay, CEA, CNRS, Institute for Integrative Biology of the Cell (I2BC), 91198, Gif-sur-Yvette, France.
